 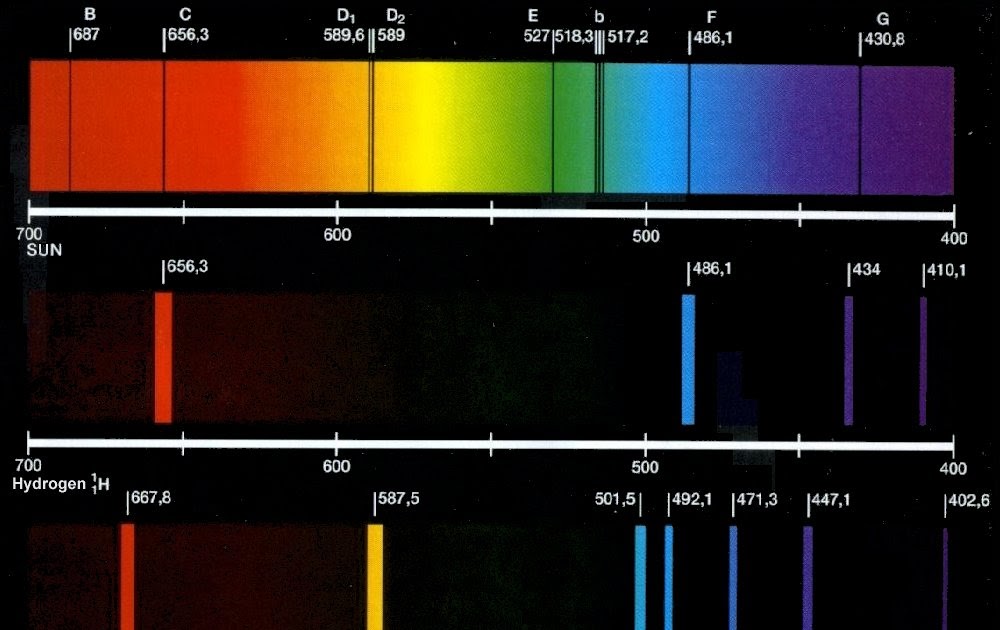
There are two other blue lines at 404.656 nm and 407.781 nm and a weak line at 491.604 nm. Atomic Spectroscopy, submitted for publication (2004). The prominent mercury lines are at 435.835 nm (blue), 546.074 nm (green), and a pair at 576.959 nm and 579.065 nm (yellow-orange). The wavelengths, intensities, and spectrum assignments are given in a table for each element, and the data for the approximately 12,000 lines of all elements are also collected into a single table, sorted by wavelength (a 'finding list'). The conditions employed to prevent the molecules being broken up were : feeble electrical. At the right of the image are the spectral lines through a 600 line/mm diffraction grating. A second emission spectrum of S02 was investigated by Miss Lowater. Such signs are excited by voltages of a few thousand volts produced by a transformer that raises the voltage of the ordinary AC line voltage.Īt left is a mercury spectral tube excited by means of a 5000 volt transformer. NIST Physical Measurement Laboratory, Handbook of Basic Atomic Spectroscopic Data. This is a section of the sign shown below, which has a central neon section and another gas mixture producing blue light around it. Then the image below was reduced and superimposed on the image above, because with the exposure reasonable for the bright tube, only the red lines were visible on the photograph. Click on the name in this column to launch the Applet which displays an emission line spectrum of the. The image below is composed of segments of three photographs to make the yellow and green lines more visible along with the much brighter red lines. All data are taken from the NIST Atomic Spectra. Again, at 130.2 nm, three overlapping oxygen emission lines are visible. 
The emission lines at 141.2, 149.3, and 174.3 nm are attributed to atomic nitrogen as the argon measurements could not be conducted in controlled atmosphere. This is an attempt to give a reasonable accurate picture of the appearance of the neon spectrum, but both the images are composite images. In addition to the argon emission, atomic emission lines originating from impurities are observable. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
